Despite treatment advances, prognosis remains poor for adults
with R/R B-cell ALL1,2
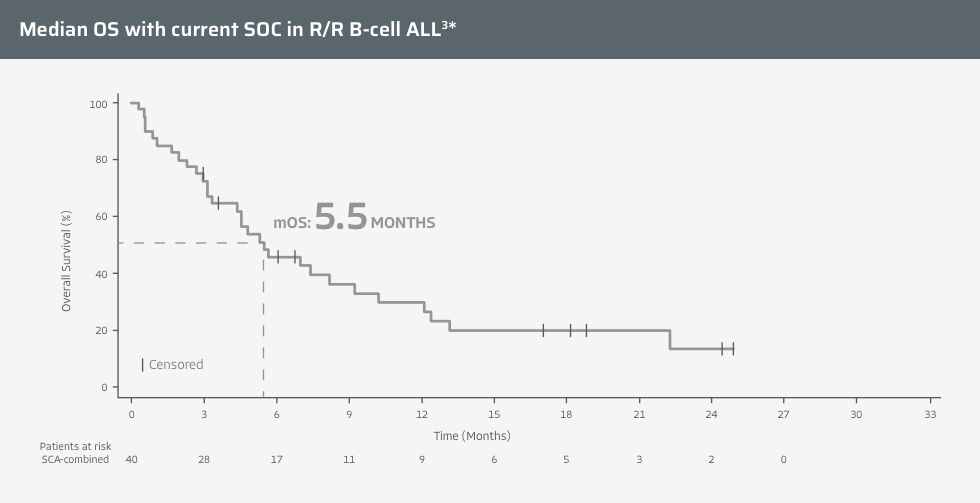
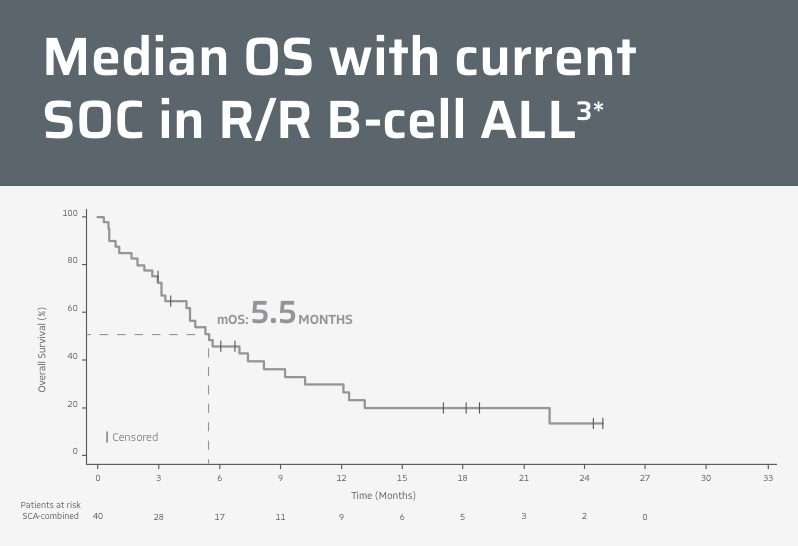
Regardless of prior immunotherapy, median OS is <6 months with SOC therapies in historical clinical trials3
Pinch to zoom
Complete response rates are also low with standard R/R ALL therapies2-4


*Data from a 2021 retrospective cohort study of efficacy and safety of treatments considered to be the SOC in adult patients with R/R ALL. The study included three patient-matched historical control cohorts for a SCA-combined population that included patients with and without prior exposure to blinatumomab or inotuzumab ozogamicin. The primary endpoint was the CR/CRi rate of the SCA-1 population (naïve to blinatumomab and inotuzumab ozogamicin).3
†Data from a pooled analysis of adult patients with R/R Ph-negative, B-precursor ALL across 11 groups or sites in the United States and Europe. 2210 salvage records were included in total, with 1628 patients in first salvage, 374 patients in second salvage, and 160 patients in third or greater salvage.4
‡Data from a review of 245 patients with ALL treated at the University of Texas MD Anderson Cancer Center who received first salvage therapy following either primary treatment failure following induction or for relapsed ALL <12 months after initial therapy.2
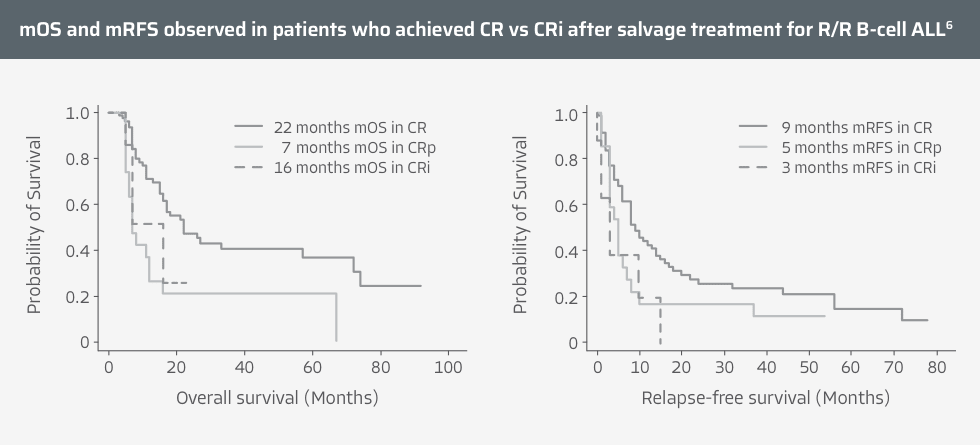
Type of response is critical for predicting outcomes in R/R B-cell precursor ALL5,6
10% to 40% of patients with R/R ALL go on to receive allo-SCT5
- In a retrospective analysis of 180 adults who received allo-SCT, 86% achieved a CR one month after allo-SCT and mOS was 23 months at a median study follow-up of 10.2 years7
- Achieving a CR before allo-SCT is a favorable prognostic factor7
CR is associated with improved outcomes in adults with R/R B-cell ALL6
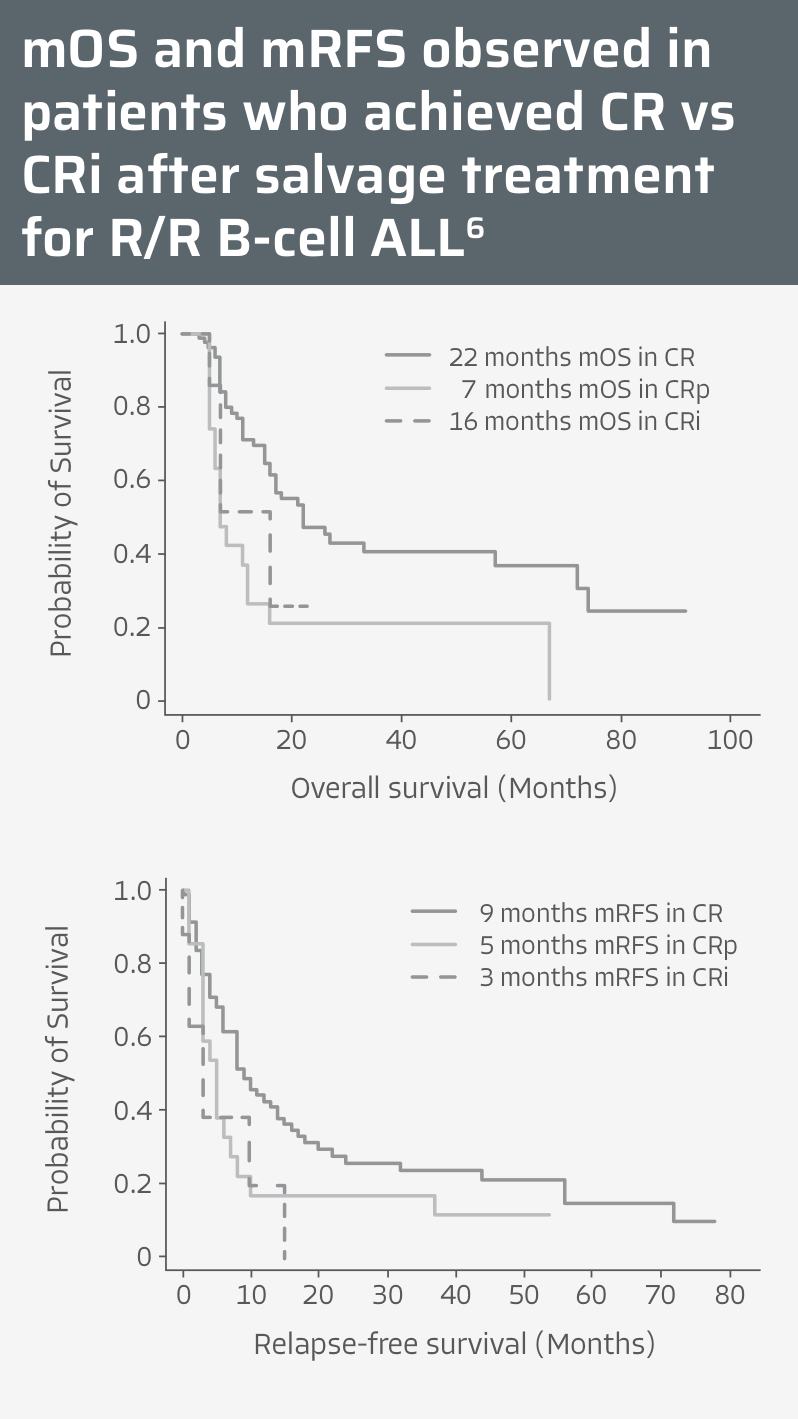
Pinch to zoom
- Along with CR, MRD-negative status may be a major determinant for better mRFS in adult patients with R/R B-cell ALL6
There is an urgent need for treatments that can improve outcomes by
delivering higher rates of CR in adult patients with R/R B-cell ALL4,8
ALL=acute lymphoblastic leukemia; allo-SCT=allogeneic stem cell transplant; CR=complete remission; CRi=complete remission with incomplete hematologic recovery; CRp=complete remission with incomplete platelet counts; mOS=median overall survival; MRD=minimal residual disease; mRFS=median relapse-
free survival; OS=overall survival; Ph=Philadelphia chromosome; R/R=relapsed or refractory; SCA=synthetic control arm; SOC=standard of care.
References: 1. Gökbuget N, Stanze D, Beck J, et al. Outcome of relapsed adult lymphoblastic leukemia depends on response to salvage chemotherapy, prognostic factors, and performance of stem cell transplantation. Blood. 2012;120(10):2032-2041. 2. Kantarjian HM, Thomas D, Ravandi F, et al. Defining the course and prognosis of adults with acute lymphocytic leukemia in first salvage after induction failure or short first remission duration. Cancer. 2010;116(24):5568-5574. 3. Shah BD, Ghobadi A, Oluwole OO, et al. Two-year follow-up of KTE-X19 in patients with relapsed or refractory adult B-cell acute lymphoblastic leukemia in ZUMA-3 and its contextualization with SCHOLAR-3, an external historical control study. J Hematol Oncol. 2022;15(1):170. 4. Gökbuget N, Dombret H, Ribera JM, et al. International reference analysis of outcomes in adults with B-precursor Ph-negative relapsed/refractory acute lymphoblastic leukemia. Haematologica. 2016;101(12):1524-1533. 5. Craddock C, Hoelzer D, Komanduri KV. Current status and future clinical directions in the prevention and treatment of relapse following hematopoietic transplantation for acute myeloid and lymphoblastic leukemia. Bone Marrow Transplant. 2019;54(1):6-16. 6. Saygin C, Papadantonakis N, Cassaday RD, et al. Prognostic impact of incomplete hematologic count recovery and minimal residual disease on outcome in adult acute lymphoblastic leukemia at the time of second complete response. Leuk Lymphoma. 2018;59(2):363-371. 7. Greil C, Engelhardt M, Ihorst G, et al. Prognostic factors for survival after allogeneic transplantation in acute lymphoblastic leukemia. Bone Marrow Transplant. 2021;56(4):841-852. 8. Fulcher J, Leung E, Christou G, Bredeson C, Sabloff M. Selecting the optimal targeted therapy for relapsed B-acute lymphoblastic leukemia. Leuk Lymphoma. 2020;61(9):2271-2273.